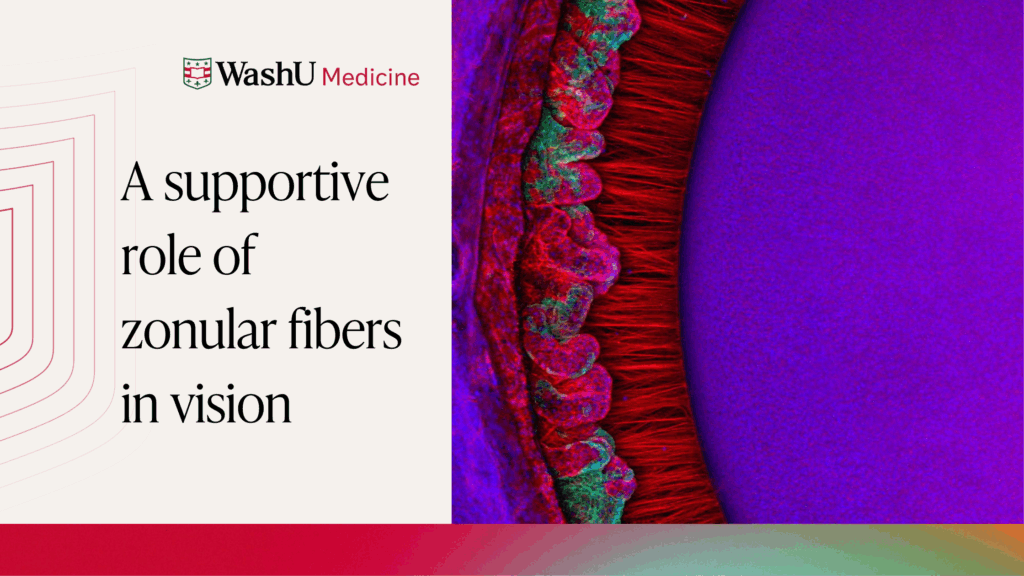
This Research Insight covers a publication from the Bassnett Lab. Here, we highlight how findings by Pooja Rathaur, PhD and colleagues have contributed to our understanding of how the fibrous molecules that comprise the zonular fibers of the eye support vision.
In their paper published in Cells, scientists in the lab of Steven Bassnett, PhD—professor of ophthalmology at WashU Medicine—review the structure and composition of zonular fibers and present new evidence of how their structure enables their function.

Here, Pooja Rathaur, PhD—a postdoctoral fellow in the Bassnett Lab—and colleagues manipulated zonular fibers in the mouse eye to test the physical limits of their ability to stretch. Their experiments provide new insights into key molecules that contribute to the tensile strength of zonular fibers and their ability to support the optical components of the eye.
Zonular fibers play both structural and functional roles in vision
Clear vision depends on the ability of the lens to focus light on the light-sensitive retina. This ability is assisted by zonular fibers, which anchor, center, and suspend the lens in the eye by connecting it to the ciliary muscle.
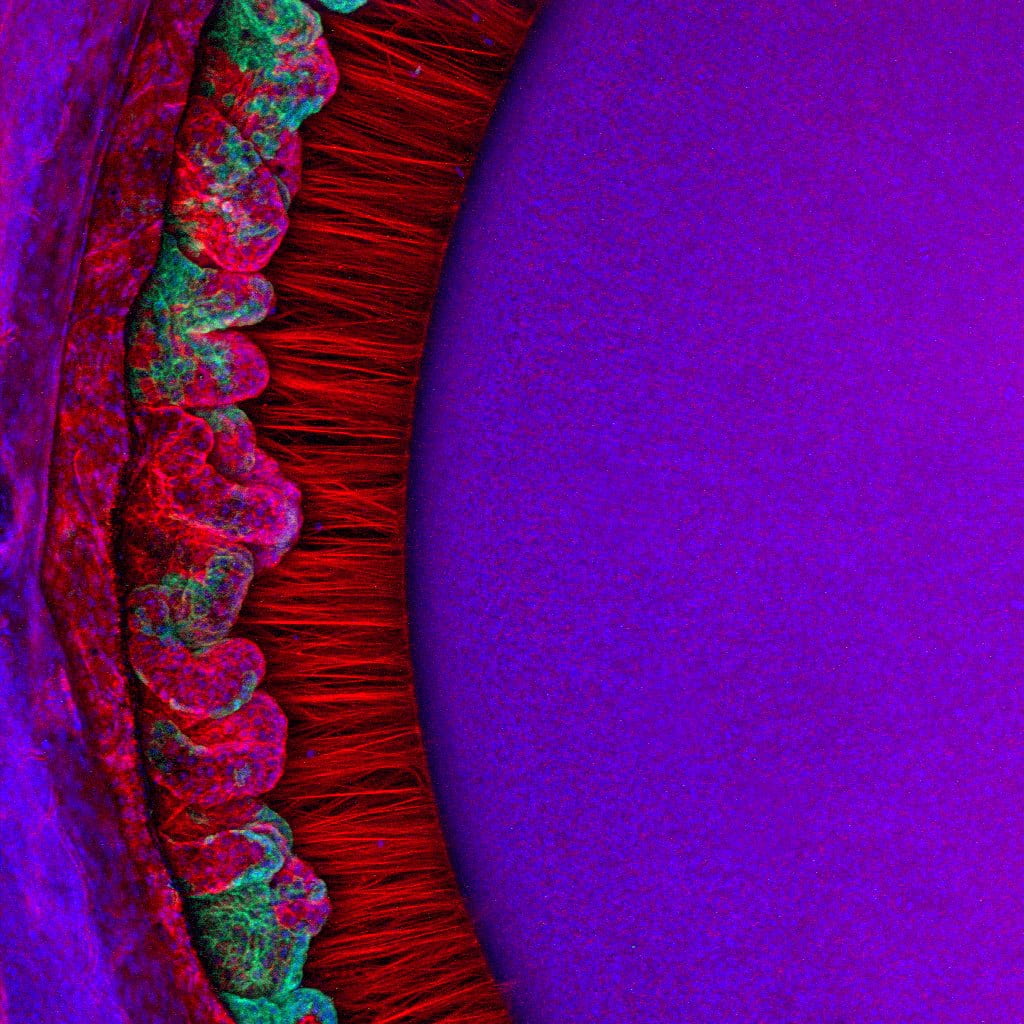
Some species—such as humans—can shift their vision rapidly between near and distant objects. In these cases, zonular fibers have the additional role of transmitting force from the ciliary muscle to the lens in a dynamic process called accommodation. During this process, the ciliary muscle relaxes to focus vision distantly, causing the zonular fibers to pull the lens into a flattened conformation and changing how light is focused on the retina. In contrast, the ciliary muscle contracts to focus vision close-up, allowing the lens to return to its rounded shape.
In both non-accommodating and accommodating species—like the mouse and human, respectively—the structural integrity of zonular fibers is crucial to support vision.
The composition of zonular fibers
Zonular fibers differ considerably between species in their structure and number. Accommodating eyes—like those of the human—typically have fewer zonular fibers that are thicker than those found in non-accommodating eyes, like those of the mouse. Despite these structural differences, similarities have been reported in their protein composition across species.
Across species, zonular fibers are predominantly composed of microfibril proteins—such as fibrillin-1 and latent TGFb-binding protein-2 (LTBP2). One notable exception is the lysyl oxidase-like 1 (LOXL1) protein, which might interact with or regulate these microfibril proteins. The genes that encode these proteins have been implicated in diseases in which patients often experience destabilization of the lens and/or iris due to failure of the stabilizing zonular fibers.
Disentangling the biomechanics of zonular fibers
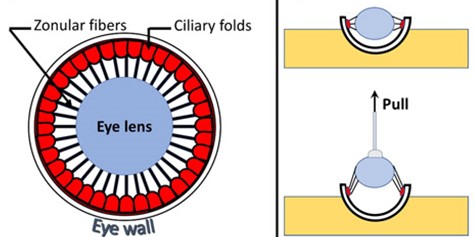
Rathaur and colleagues turned to the mouse eye to interrogate how specific genes implicated in human diseases might destabilize zonular fibers and lead to ocular symptoms, including lens dislocation.
They deleted genes of interest from the mouse eye and assessed the resulting tensile strength of the zonular fibers. To do so, Rathaur and colleagues slowly pulled on the lens while measuring the force that the zonular fibers could withstand and the distance they could stretch before breaking. Their results point toward putative disease mechanisms and give insights into potential therapeutic targets that may be explored in future studies.
The role of microfibril proteins in zonular fiber stability
The Bassnett Lab previously reported that deletion of fibrillin-1 from the mouse eye resulted in fewer and thinner zonular fibers, which were unable to effectively support the lens in the eye. In this study, Rathaur and colleagues refined this experiment by employing a targeted approach to specifically test a mutation of fibrillin-1 that has been linked to Marfan syndrome in humans. They found that the zonular fibers of mice carrying this human mutation were considerably weaker than those of mice without the mutation, despite maintaining their ability to center the lens in the eye.
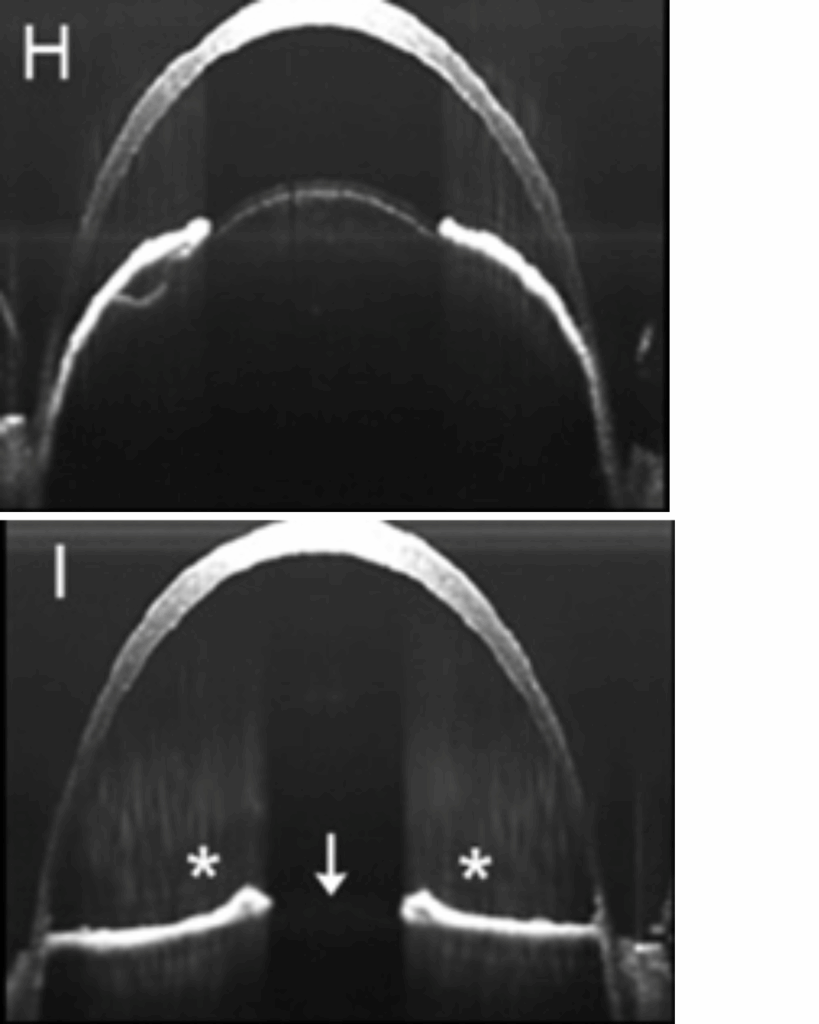
Rathaur and colleagues also observed decreased strength of zonular fibers in the eyes of mice lacking LTBP2, which has been linked to Weill-Marchesani Syndrome-3, in which patients frequently experience a dislocated lens. In this case, however, the weakening of zonular fibers appeared to be progressive. The zonular fibers of mice lacking LTBP2 were comparable to control at 1 month, but began weakening by 2 months, leading to complete lens dislocation by 6 months.
Together, these results highlight the key role of microfibril proteins for maintaining the structural integrity of zonular fibers and supporting their function in stabilizing the lens in the eye.
An unexpected role for a cross-linking enzyme
Finally, Rathaur and colleagues explored the role of LOXL1 in zonular fibers, as it has been reported at unexpectedly high levels in the zonule, despite the absence of the proteins it typically interacts with. LOXL1 mutations have been linked to exfoliation syndrome, which can manifest ocular symptoms including lens dislocation.
Surprisingly, when Rathaur and colleagues deleted LOXL1 from the mouse eye, they observed a progressive strengthening of zonular fibers compared to mice with LOXL1 present. Moreover, they found that the benefit conferred by loss of LOXL1 was sufficient to rescue the deficit incurred by loss of the microfibril protein fibrillin-1. Together, these findings suggest that LOXL1 might serve a novel regulatory function in zonular fibers that can influence its structure and stability.
Future studies are needed to identify the precise function of LOXL1 in the zonular fibers and to understand the biology that drives the unexpected functional rescue observed by the combination of LOXL1 and fibrillin-1 deletions in the mouse eye. However, Rathaur and colleagues’ findings highlight a unique opportunity to further explore disease mechanisms that lead to the structural failure of zonular fibers and to innovate potential treatment strategies to strengthen them.