Autophagy Regulates Müller Glial Cell Inflammatory Activation
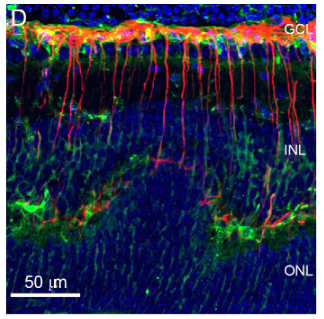
A new study published in Investigative Ophthalmology & Visual Science highlights the critical role of autophagy—the body’s process of recycling proteins—in regulating inflammation within the retina. The research, led by Thomas Ferguson, PhD, and Philip Ruzycki, PhD, from the John F. Hardesty, MD, Department of Ophthalmology & Visual Sciences at WashU Medicine, sheds new light on how the eye preserves its delicate balance between defense and destruction.
Thomas Ferguson, PhD is widely recognized as a pioneer in understanding the eye’s remarkable ability to block harmful immune responses, a process known as “immune privilege.” Neural tissues, such as the eye and the retina, must avoid unchecked inflammation and immunological responses or risk irreparable damage and loss of vision.

In this study, Ferguson and Ruzycki used animal models and single-cell RNA sequencing to investigate the role of Müller glial cells—specialized support cells in the retina—in maintaining immune privilege. Their findings revealed that when Müller glia lose the ability to perform autophagy (recycling their own proteins), they become persistently activated. This hyperactivation triggers stronger immune responses, ultimately leading to tissue damage and vision loss.
Thomas Ferguson, PhD explained:
“Our studies add another player to the complex processes that make up immune privilege, which has been called ‘nature’s protective eyewear.’”
Co-author Philip Ruzycki, PhD, added:
“Our research demonstrates that Müller glia are not passive support cells, but active regulators of the retinal immune environment. When their ability to maintain homeostasis is compromised, the consequences can be devastating for vision.”

Philip Ruzycki, PhD
Assistant Professor, Ophthalmology and Visual Sciences
- Email: p.ruzycki@nospam.wustl.edu
This discovery provides a new understanding of how immune privilege in the eye is maintained and disrupted, offering potential new targets for therapies aimed at preventing or treating inflammatory retinal diseases.
Read the full study here: Autophagy Regulates Müller Glial Cell Inflammatory Activation
Read more about the research of the Ferguson Lab.
About WashU Medicine
WashU Medicine is a global leader in academic medicine, including biomedical research, patient care and educational programs with 2,900 faculty. Its National Institutes of Health (NIH) research funding portfolio is the second largest among U.S. medical schools and has grown 56% in the last seven years. Together with institutional investment, WashU Medicine commits well over $1 billion annually to basic and clinical research innovation and training. Its faculty practice is consistently within the top five in the country, with more than 1,900 faculty physicians practicing at 130 locations and who are also the medical staffs of Barnes-Jewish and St. Louis Children’s hospitals of BJC HealthCare. WashU Medicine has a storied history in MD/PhD training, recently dedicated $100 million to scholarships and curriculum renewal for its medical students, and is home to top-notch training programs in every medical subspecialty as well as physical therapy, occupational therapy, and audiology and communications sciences.